Allakos Announces Initiation of Phase 2b Clinical Trial of Subcutaneous Lirentelimab in Chronic Spontaneous Urticaria (CSU)
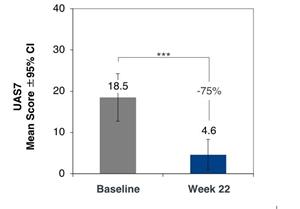
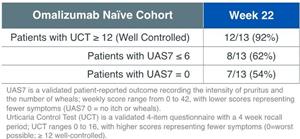
The Phase 2b CSU clinical trial follows positive results from an open-label Phase 2a clinical trial with intravenous lirentelimab in patients with chronic urticaria, including cohorts of patients with omalizumab naïve CSU and omalizumab refractory CSU (link). Patients in the omalizumab naïve cohort had urticaria symptoms despite treatment with antihistamines (up to 4x labeled dose) and had not been previously treated with omalizumab. Omalizumab naïve patients receiving monthly lirentelimab for 6 doses reported a 75% improvement in the 7 day urticaria activity score (UAS7) with 92% of patients achieving a urticaria control test (UCT) score ≥ 12.
Figure 1: Change from baseline in UAS7 in omalizumab naïve cohort (n=13)
Figure 2: UCT and UAS7 response in omalizumab naïve cohort (n=13)
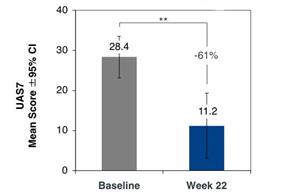
Patients in the omalizumab refractory cohort had urticaria symptoms despite treatment with antihistamines (up to 4x labeled dose) and treatment with up to 600 mg (2x highest labeled dose) omalizumab for an average duration of 10 months. Omalizumab refractory patients receiving monthly lirentelimab for 6 doses reported a 61% improvement in UAS7 with 57% of patients achieving a UCT score ≥ 12.
Figure 3: Change from baseline in UAS7 in omalizumab refractory cohort (n=7)
The safety results of the trial were generally consistent with previously reported intravenous lirentelimab clinical trials. The most common adverse event was mild to moderate infusion-related reactions (such as flushing, feeling of warmth, headache, nausea, and dizziness) which occurred mostly during the first infusion. Full results of the trial were published in
Phase 2b Chronic Spontaneous Urticaria Trial Design (MAVERICK)
The Phase 2b clinical trial is a 12-week, multicentered, randomized, double-blind, placebo-controlled trial that will enroll approximately 110 adult patients with antihistamine refractory chronic spontaneous urticaria (including patients with prior biologics treatment). Patients will be randomized 1:1 to receive doses of subcutaneous lirentelimab 300mg or placebo once every two weeks (Q2W). The primary end point of the trial is the change from baseline in UAS7 at 12 weeks.
About Chronic Spontaneous Urticaria (CSU)
CSU is a complex, chronic inflammatory disease considered to be primarily driven by mast cells. However, recent evidence suggests that eosinophils may also have a role in symptomology. CSU is an often-debilitating skin condition characterized by frequent and unpredictable eruption of hives, severe itching and swelling. The disease affects up to 3.5 million patients in the
Allakos Development Programs
About
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 as contained in Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Such forward-looking statements include, but are not limited to, Allakos’ progress, business plans and areas of focus, the expected timing of reporting topline data from its Phase 2 and 2b clinical trials of lirentelimab, the advancement of IND enabling studies for AK006 and initiation of a Phase 1 clinical trial with AK006. Such statements are subject to numerous important factors, risks and uncertainties that may cause actual events or results to differ materially from current expectations and beliefs, including but not limited to: Allakos’ stages of clinical drug development; Allakos’ ability to timely initiate clinical trials for AK006; Allakos’ ability to obtain required regulatory approvals for its clinical trials; uncertainties related to the enrollment of patients in its clinical trials; Allakos’ ability to demonstrate sufficient safety and efficacy of its product candidates in its clinical trials; uncertainties related to the success of later-stage clinical trials, regardless of the outcomes of preclinical testing and early-stage trials; Allakos’ ability to obtain regulatory approvals to market its product candidates; market acceptance of Allakos’ product candidates; uncertainties related to the projections of the size of patient populations suffering from the diseases
Source:
1 Altrichter S et al. J Allergy Clin Immunol. 2022 Sep;150(3):736-737
2 Maurer M et al. Allergy. 2011 Mar;66(3):317-30
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/912f186d-d61e-4b00-91e9-33aa17cb6443
https://www.globenewswire.com/NewsRoom/AttachmentNg/dfc68b6e-5bf0-4d19-bc2c-67698da9f357
https://www.globenewswire.com/NewsRoom/AttachmentNg/86fd042d-f180-4bd3-ad21-bcc287c5848e

Investor Contact:Adam Tomasi , President and COOAlex Schwartz , VP Strategic Finance and Investor Relations ir@allakos.com Media Contact:Denise Powell denise@redhousecomms.com
Source: Allakos Inc.